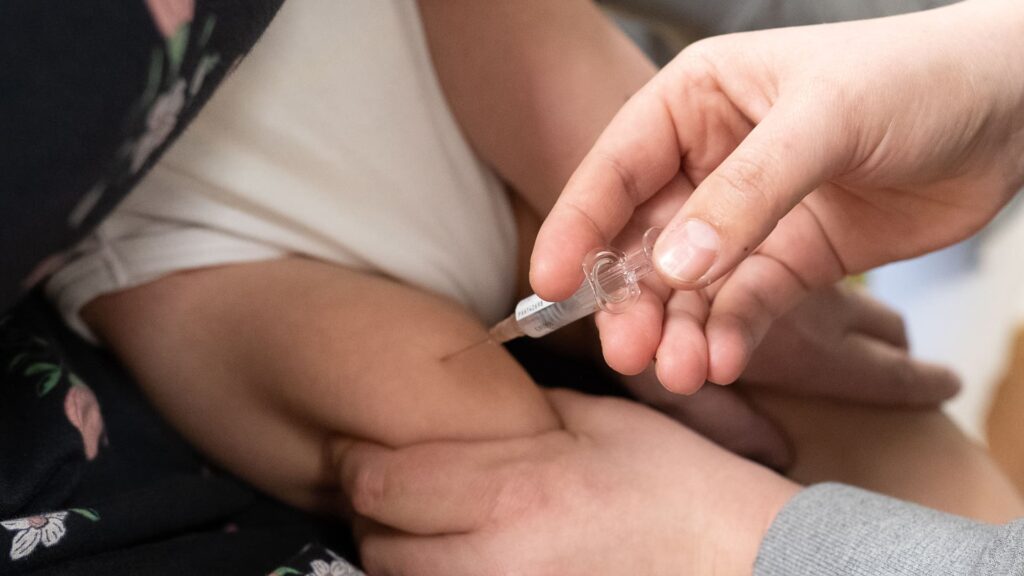
A doctor vaccinates an infant against respiratory syncytial virus (RSV) in a treatment room of her paediatric practice.
Swen Pförtner | Picture Alliance | Getty Images
The Centers for Disease Control and Prevention’s independent panel of advisors on Friday recommended Pfizer‘s maternal vaccine that protects infants from respiratory syncytial virus, the leading cause of hospitalization among babies in the U.S.
Eleven advisors recommended that expectant mothers 32 to 36 weeks into their pregnancy receive the shot during September through January to protect their children from RSV. One advisor voted against the recommendation.
CDC Director Mandy Cohen needs to sign off on that recommendation before the shot can be administered to mothers. Pfizer’s vaccine, called Abrysvo, is already approved and available in the U.S. for adults ages 60 and up.
The panel’s recommendation puts Pfizer’s maternal shot on track to reach the public as RSV begins to spread at higher levels. Public health officials hope the vaccine and other treatments will make the U.S. more equipped to combat the virus this fall and winter, especially after the nation faced an unusually severe RSV season last year.
RSV usually causes mild, cold-like symptoms. But younger children and older adults are particularly vulnerable to more severe RSV infections.
Each year, the virus kills a few hundred children younger than 5, and 6,000 to 10,000 seniors, according to the CDC. RSV also causes around 58,000 to 80,000 hospitalizations among children younger than 5 years old each year, the CDC said.
Pfizer has said its maternal vaccine could prevent up to 16,000 hospitalizations and more than 300,000 visits to the doctor due to RSV if the shot becomes available in the U.S. this fall and winter.
“This fall marks the start of the annual respiratory infection season in the Northern Hemisphere, and we are prepared with vaccines against multiple infectious diseases and – for the first time in history – an available RSV vaccine to help prevent disease in two at-risk populations,” said Dr. Luis Jodar, Pfizer’s chief medical officer for vaccines medical development.
The company’s single-dose vaccine is the first RSV treatment to use maternal immunization: Expectant mothers get vaccinated with the shot, which triggers antibodies that are passed to the fetus. That provides infants with protection against the virus from birth through the first six months of life.
The Food and Drug Administration established the 32- to 36-week dosing timeframe when it approved the shot.
The FDA and CDC cleared an RSV antibody injection for infants from Sanofi and AstraZeneca over the summer, but that treatment is administered directly to babies. The CDC recommended the injection, known as Beyfortus, to all infants under 8 months of age and some older babies.
A subgroup of the CDC advisory panel “felt strongly” that most infants won’t need both Beyfortus and Pfizer’s Abrysvo, CDC medical officer Dr. Jefferson Jones said during the advisory meeting Friday. That subgroup reviews published and unpublished data to develop recommendation options for the panel.
“The pregnant person and their prenatal care provider will need to make the decision during pregnancy regarding which RSV prevention product to use,” said Jones.
Weighing Abrysvo’s efficacy and safety data
The CDC panel’s recommendation of Pfizer’s maternal vaccine was based on data from a phase three trial on nearly 7,400 participants. But mothers received the shot 24 to 36 weeks into their pregnancy in the trial, which is a wider period than the approved dosing time frame.
During the first 90 days after birth, the shot was nearly 82% effective at preventing severe RSV disease in newborns and 57% effective at keeping babies from needing a doctor’s visit due to RSV-related breathing problems.
That efficacy appeared to lower slightly over time: By six months after birth, Pfizer’s shot was about 70% effective at preventing severe disease and 51% effective at avoiding a trip to the doctor.
A panel of advisors to the FDA generally praised the efficacy of Pfizer’s maternal vaccine but expressed concerns about potential safety risks.
In the phase three trial, a slightly higher number of premature births occurred among mothers who took the shot compared to those who received a placebo: 5.7% versus 4.7%, respectively.
Pfizer, the FDA and CDC staff have said the difference wasn’t statistically significant.
Pfizer has also sent the CDC advisory panel unpublished data suggesting that the rate of preterm births decreased for women who only received the shot during the approved dosing time frame of 32 to 36 weeks into pregnancy, Dr. Katherine Fleming-Dutra, a pediatrician with the National Center for Immunization at the CDC, said during the advisory meeting on Friday.
Fleming-Dutra said 4.2% of births were premature among mothers who took the shot during that time frame compared with 3.7% among those who received a placebo.
“The rate of preterm birth decreased as there’s less opportunity to be born preterm and also the imbalance between vaccine and placebo groups narrowed with the approved dosing interval,” Fleming-Dutra said.
Still, the prescribing label for Pfizer’s vaccine will come with a warning not to administer the shot before 32 weeks of pregnancy because of that “numerical imbalance” in premature births, the FDA said in its approval.
The FDA is requiring Pfizer to examine the risk of premature births in a post-marketing study on the vaccine. Post-marketing refers to research conducted on a product after it receives FDA approval.
Pfizer’s examination will also involve evaluating any pregnancy-related complications following vaccination, Alejandra Gurtman, the company’s senior vice president of clinical research and development for vaccines, told CNBC last month.
That includes eclampsia, which refers to seizures that develop during pregnancy or shortly after birth.
Pfizer will launch a pregnancy registry that will allow women and obstetricians to call and report any adverse events after patients receive the vaccine, according to Gurtman.
Read the full article here